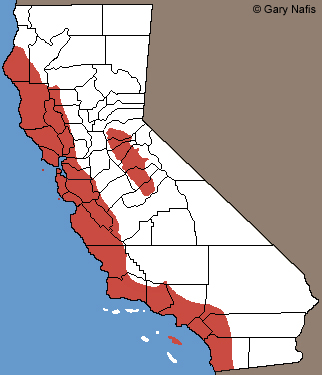
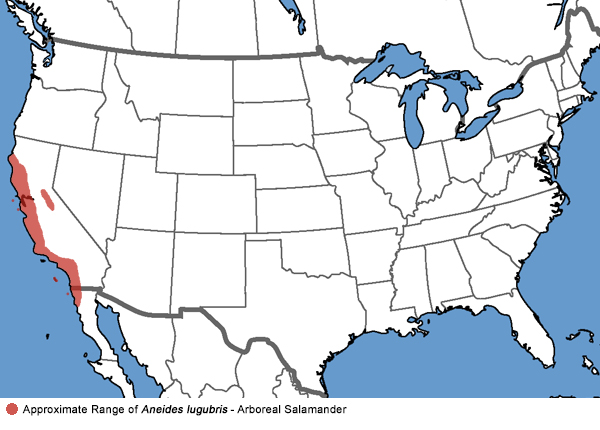
Arboreal Salamander - Aneides lugubris
(Hallowell, 1849)Description • Taxonomy • Species Description • Scientific Name • Alt. Names • Similar Herps • References • Conservation Status
 |
|||||||||||||||||||||||||||||||||||||||||||||||||||
| Adult with small spots, Contra Costa County | |||||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| Adult with small spots, Contra Costa County | Adult with small spots, Alameda County |
Adult with small spots, Santa Cruz County |
|||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| Adult with small spots, Contra Costa County (missing the tip of its tail.) | |||||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| Adult with small spots, Contra Costa County | Adult, found on a building in downtown San Francisco, San Francisco County © Jackson Shedd |
||||||||||||||||||||||||||||||||||||||||||||||||||
 |
|||||||||||||||||||||||||||||||||||||||||||||||||||
| Adult with large spots, Monterey County | |||||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| Adult male with large spots, Monterey County coastal sand dunes | Adult male with large spots, Monterey County coastal sand dunes | ||||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| Adult active at night in situ, Marin County |
Adult active at night in situ, Marin County |
Adult active at night in situ, Marin County |
Adult active at night in situ, Marin County |
||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| Adult in situ emerging at night from a rock crevice, Marin County | Adult male with large head scar, Marin County | Adult, Santa Clara County © aquariumkids.com This large adult salamander crawled under the crack of the front door of a house on a rainy night in January. |
|||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| Adult with few small spots, from Mendocino County |
Adult, Anchor Bay, Mendocino County © Richard L. P. Custer |
Adult, Stanislaus County © Jared Heald | This adult was rescued from the bottom of the deep end of a swimming pool in San Luis Obispo County. © Laurie Dey | ||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| Adult, Alameda County | Adult, San Mateo County © Jared Heald | Adult with small spots, found under a garbage can after the rains started in October in Santa Clara County. © John Roberts |
|||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| Adult, San Luis Obispo county, © Andrew Harmer |
Adult, San Luis Obispo County © Henk Wallays |
Adult with small spots, San Diego County © Paul Maier | Adult, Orange County © Jason Jones | ||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| Two Adults found in the Santa Ana Mountains, Orange County © Cooper Bailey | Adult, Los Angeles County © Gregory Litiatco |
Adult with small spots, San Diego County © Stuart Young | |||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| Adults with small spots, San Diego County © Jay Keller | Adult with small spots, Orange County © Tadd Kraft |
||||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| Adult, Coastal Southern California © Ivan Vershynin | Adult with a nice city view, San Luis Obispo County © Spencer Riffle | Adult hunting in situ, San Luis Obispo County © Spencer Riffle | |||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| This adult was found on a road on a rainy night in a surprisingly dry, treeless area in the foothills of far eastern Alameda County © Noah Morales | Adult, western Stanislaus County © Noah Morales |
Adult, western Stanislaus County © Noah Morales |
|||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| Adult male, Santa Monica Mountains, Los Angeles County © Hugo Rojas | Adult, Sierra Nevada Mountains, Tuolumne County © Rick Staub | Subadult from the Santa Monica Mountains, Los Angeles County © Max Roberts |
Adult with heavy spotting, Contra Costa County © Max Roberts |
||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| Adult, Los Angeles County © Max Roberts | Adult with minimal spotting, found at the entrance of a crevice in a rock face (right) in Los Angeles County © Max Roberts |
||||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| Adult climbing a Coast Live Oak (Quercus agrifolia) in Los Angeles County © Max Roberts | Adult in rock crevice in Los Angeles County © Max Roberts | Adult, northern San Diego County © Tadd Kraft |
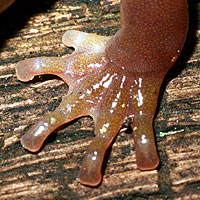
Arboreal Salamanders have toes with expanded tips that help them climb vertical surfaces like tree trunks and cliffs. |
||||||||||||||||||||||||||||||||||||||||||||||||
| Arboreal Salamanders from Southeast Farallon Island (Formerly known as Aneides lugubris farallonensis) |
|||||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
|||||||||||||||||||||||||||||||||||||||||||||||||
| Adult with large spots Southeast Farallon Island © Oscar Johnson | Adult with very large spots, Southeast Farallon Island © Morgan Stickrod | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Juveniles | |||||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| Juvenile, Santa Cruz County | Hatchling, Marin County | Hatchling, San Luis Obispo County © Spencer Riffle |
Hatchling, San Luis Obispo County © Spencer Riffle |
||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| Juvenile, Marin County | Juvenile, Santa Cruz County © Mark Gary |
Hatchling, San Luis Obispo County © Spencer Riffle |
Juvenile, Santa Cruz County © Brad Alexander |
||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
|||||||||||||||||||||||||||||||||||||||||||||||||
| Hatchling, San Luis Obispo County © Spencer Riffle |
Juvenile, Calaveras County © Noah Morales |
Bluish juvenile, found on a rocky slope at about 3000 ft. elevation in Mariposa County © Adam Gitmed | |||||||||||||||||||||||||||||||||||||||||||||||||
 |
|||||||||||||||||||||||||||||||||||||||||||||||||||
| Yearling, Los Angeles County © Max Roberts | |||||||||||||||||||||||||||||||||||||||||||||||||||
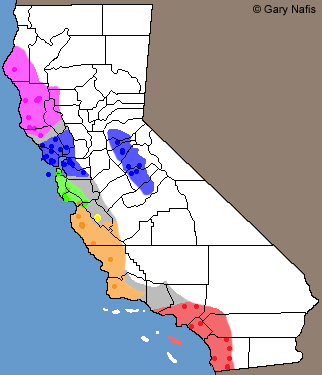
| Comparisons with Sympatric Aneides niger - Santa Cruz Black Salamander | |||||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||||
| Top: Head of adult A. niger Bottom: Head of adult A. lugubris © Ryan Sikola |
Top: Head of juvenile A. lugubris Bottom: Head of juvenile A. niger © Ryan Sikola |
||||||||||||||||||||||||||||||||||||||||||||||||||
| Male Combat and Defense | |||||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
|||||||||||||||||||||||||||||||||||||||||||||||||
| Two adult males locked in bloody combat in Santa Cruz County © Sasha Robinson. The picture on the far right was taken about an hour after the first two pictures and they were still fighting. | |||||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| Be careful when handling this salamander! It has lots of sharp teeth and isn't afraid to use them. Mike Spencer found it, Val Johnson took the pictures, and Shannon Hoss lost some blood when she picked up this toothy beast in Mendocino County. © Val Johnson |
Adult, Santa Barbara County © Ryan Sikola This tough survivor is missing an eye and part of its face, but still appears healthy. Meles compete in violent combat, which might have produced this injury, but it could also be the result of an encounter with a predator. |
||||||||||||||||||||||||||||||||||||||||||||||||||
| Arboreal Salamanders with an Aberrant or Unusual Appearance | |||||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| Top: Typical Arboreal Salamander. Bottom: Albino or non-pigmented adult Arboreal Salamander. Both were found in the same general location in Contra Costa County. © 2006 John Schilling |
Unusually pale adult, Marin County © Andre Giraldi This salamander is much lighter in color than a typical Arboreal Salamander. Other typically colored salamanders were found near it. It could be missing some of its melanin or dark pigment, or its appearance could be a result of the salamander changing its color, which was described by Robert Stebbins. See description below. |
||||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| These unusually-pigmented adults were found together in San Luis Obispo County - Unspotted (left) Leucistic (right) © Ryan Sikola |
Leucistic adult, San Luis Obispo County © Ryan Sikola | Adult with no light spotting, San Luis Obispo County © Ryan Sikola |
This adult from Santa Clara County has a mottled head. © Katherine Hughes |
||||||||||||||||||||||||||||||||||||||||||||||||
Short Video |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| A short video and photos showing a possibly leucistic young adult Arboreal Salamander discovered walking at night in the mountains of Tuolumne County at about 4,975 ft. (1,516 m.) elevation. which is almost at the current elevation record of 5,000 ft. © Connor Stone | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Aestivation and Egg Brooding | |||||||||||||||||||||||||||||||||||||||||||||||||||
| During the dry period, usually from May or June to October or November, Arboreal Salamanders hide out in cool moist areas, usually underground, sometimes in tree hollows, sometimes in basements and underground garages. Females lay their eggs in late Spring and early Summer and they are sometimes found guarding their eggs along with other aestivating salamanders throughout the heat of summer until the rains begin and the eggs hatch in October or November. |
|||||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| The adult Arboreal Salamander shown on the left (most likely a female) was found in an irrigation box in Sonoma County guarding a cluster of eggs attached to the box above her. The eggs were laid sometime at the beginning of July and they hatched in early October. The salamander stayed with them the entire time and continued there throughout the winter after they had hatched. The picture on the right shows the adult below a cluster of approximately ten hatchlings a day or two after hatching. They stayed in this cluster for several days. Afterwards the number of adult and juvenile salamanders observed in the box varies as they move in and out, with as many as ten observed at one time. © Sohni Yaylian | Eric Boyer discovered this large group of in situ estivating adult and juvenile Arboreal salamanders underneath some wood while remodeling a backyard in Santa Barbara County in June. © Eric Boyer |
This in situ Arboreal Salamander was found sheltering underneath the circular base of a large statue in a Santa Clara County backyard in August. © Laurie Weber |
|||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
|||||||||||||||||||||||||||||||||||||||||||||||||
| Freshly laid eggs photographed on July 7th, 2024 in the same irrigation box in Sonoma County as seen directly above from 2023 which were also laid in early July. The head of an adult guarding the eggs can also be seen, along with a juvenile that is possibly one the hatchlings from last year shown above. © Sohni Yaylian | Adult and juvenile photographed in early July, 2024 in the Sonoma County irrigation box. The juveniles could have originated as hatchlings from the eggs laid here last year shown above. © Sohni Yaylian | ||||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||||
| Old egg cluster that was found attached to the underside of a log on the ground in Contra Costa County. A hatchling was seen just below the egg cluster. © Max Roberts |
These eggs were found underneath some old redwood planks in a shaded area next to a house in Sonoma County. Though they were found near several aestivating Arboreal Salamanders in July when they lay their eggs, they are most likely the eggs of a slug or snail because of their opacity and because they don't appear to be attached to the wood. Gastropod eggs are sometimes confused for salamander eggs because of their similar size and because they are both laid in similar dark moist places. © Anonymous |
||||||||||||||||||||||||||||||||||||||||||||||||||
| Feeding | |||||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| Juvenile eating an insect larva or worm in San Luis Obispo County © Ryan Sikola | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Predation | |||||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||||
| An adult San Bernardino Ring-necked snake eating an adult Arboreal Salamander in Los Angeles County © Jonathan Benson | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Habitat | |||||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| Typical oak woodland habitat, Contra Costa County | Habitat, Contra Costa County | Arboreal salamanders inhabit this urban backyard in Oakland, Alameda County |
|||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| Coastal sand dunes habitat, Monterey County |
Rocky habitat, Marin County | Habitat at coastal marsh, San Mateo County |
Habitat, Stanislaus County © Jared Heald |
||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| Arboreal salamanders inhabit nearly-treeless, rocky, Southeast Farallon Island. | Southeast Farallon Island © Morgan Stickrod |
Southeast Farallon Island © Morgan Stickrod |
Cricket cave on Southeast Farallon Island occasionally inhabited by Arboreal Salamanders © Morgan Stickrod |
||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
| Habitat, San Diego County © Jay Keller |
Habitat, Coastal Southern California © Ivan Vershynin |
Habitat, Coastal Southern California © Ivan Vershynin |
Arboreal salamanders inhabit rocky Año Nuevo Island. |
||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||||
| A careful look underneath the fallen branches and bark of the dead tree shown above on a wet winter afternoon turned up 16 salamanders of 4 species - one Arboreal Salamander, two Coast Range Newts, one Yellow-eyed Ensatina, and 12 California Slender Salamanders, proving that wood debris on a forest floor is an important microhabitat for salamanders. Along with fallen debris, tree bark, tree cavities, root holes, and splits in trees are also useful habitat for many kinds of wildlife, including birds, mammals, amphibians and reptiles, but dead trees and their debris are often removed indiscriminately without consideration for wildlife. The Cavity Conservation Initiative is a group whose goal is to educate land managers and the public about the value of dead trees. |
|||||||||||||||||||||||||||||||||||||||||||||||||||
| Short Video and Audio | |||||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
An Arboreal Salamander resting and running. |
Several Arboreal Salamanders are seen hiding in cracks in a rock wall at night. A large one out on the rocks crawls back into a hiding spot. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Listen to this sub-adult Marin County Arboreal Salamander squeak two times. (There's a faint high-pitched squeak, then some rustling background noise, then another faint squeak.) The salamander squeaked several times when the log under which it was resting was lifted. Sound and photo © Jonathan Hakim |
An Arboreal Salamander, found sitting out on a rock at night, chirps when I grasp it across the sides firmly with my thumb and one finger. The salamander was not squeezed so hard that the sound was just the sound of air being pushed out of its lungs. This species is capable of making a fairly loud sound, but this one was very quiet, so there is a lot of background noise from a flowing creek nearby. | ||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||
|
The following conservation status listings for this animal are taken from the July 2025 State of California Special Animals List and the July 2025 Federally Listed Endangered and Threatened Animals of California list (unless indicated otherwise below.) Both lists are produced by multiple agencies every year, and sometimes more than once per year, so the conservation status listing information found below might not be from the most recent lists, but they don't change a great deal from year to year.. To make sure you are seeing the most recent listings, go to this California Department of Fish and Wildlife web page where you can search for and download both lists: https://www.wildlife.ca.gov/Data/CNDDB/Plants-and-Animals. A detailed explanation of the meaning of the status listing symbols can be found at the beginning of the two lists. For quick reference, I have included them on my Special Status Information page. If no status is listed here, the animal is not included on either list. This most likely indicates that there are no serious conservation concerns for the animal. To find out more about an animal's status you can also go to the NatureServe and IUCN websites to check their rankings. Check the current California Department of Fish and Wildlife sport fishing regulations to find out if this animal can be legally pursued and handled or collected with possession of a current fishing license. You can also look at the summary of the sport fishing regulations as they apply only to reptiles and amphibians that has been made for this website. This salamander is not included on the Special Animals List, meaning there are no significant conservation concerns for it in California according to the California Department of Fish and Game. |
||
| Organization | Status Listing | Notes |
| NatureServe Global Ranking | ||
| NatureServe State Ranking | ||
| U.S. Endangered Species Act (ESA) | None | |
| California Endangered Species Act (CESA) | None | |
| California Department of Fish and Wildlife | None | |
| Bureau of Land Management | None | |
| USDA Forest Service | None | |
| IUCN | ||
Return to the Top
© 2000 -